The killer within: inhibition of cell wall synthesis by ζ toxins.
Abstract
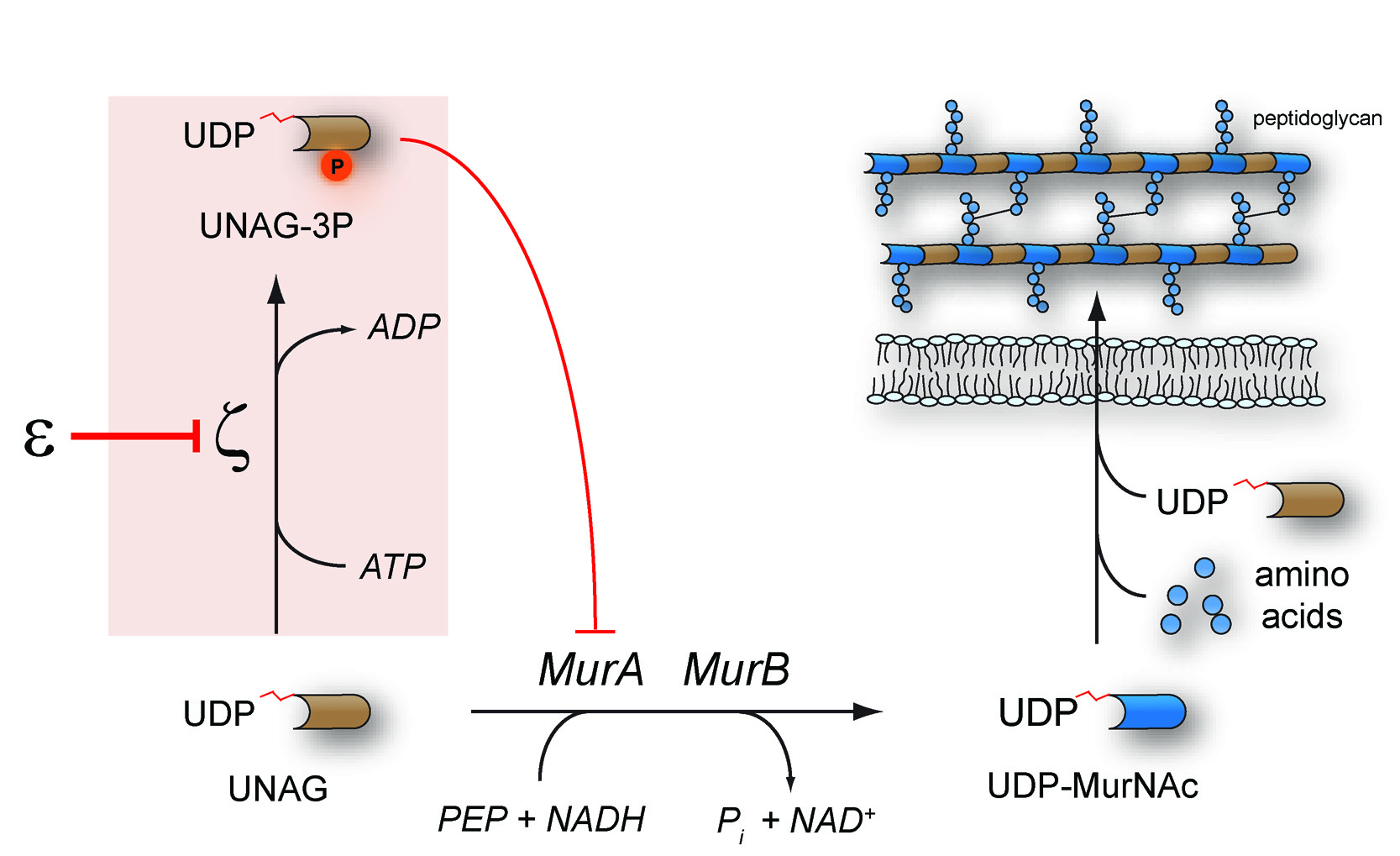
ζ toxins are bacteriotoxic proteins that are found in the genomes of various bacteria as part of ε/ζ toxin-antitoxin systems. Whereas plasmid-borne ε/ζ systems stabilize multi-resistance plasmids in pathogens, the chromosomally encoded ε/ζ homologue PezAT from Streptococcus pneumoniae was reported to enigmatically boost pneumococcal infections. However, the general mechanism of cell poisoning by ζ toxins had remained inscrutable. Therefore, we studied the phenotype of ζ toxin expression in the model organism Escherichia coli using an attenuated variant of the pneumococcal ζ toxin PezT. We observed that PezT triggers growth-rate dependent autolysis in bacteria apparently by interfering with cell wall synthesis. Further in vitro experiments revealed that members of the ζ toxin family, in general, phosphorylate the essential peptidoglycan precursor UDP-N-acetylglucosamine (UNAG). We found that the product of this reaction, UNAG-3P, inhibits the enzyme MurA, which catalyzes the conserved entry point into peptidoglycan synthesis in all bacteria. Consequently, ζ toxins define a novel family of kinases that convert an essential metabolite into a toxic compound. Our findings also suggest a mechanism that explains the role of PezT during pneumococcal infections: PezT induced autolysis in a subpopulation of cells might increase the overall infection progress through release of cytosolic virulence factors.